 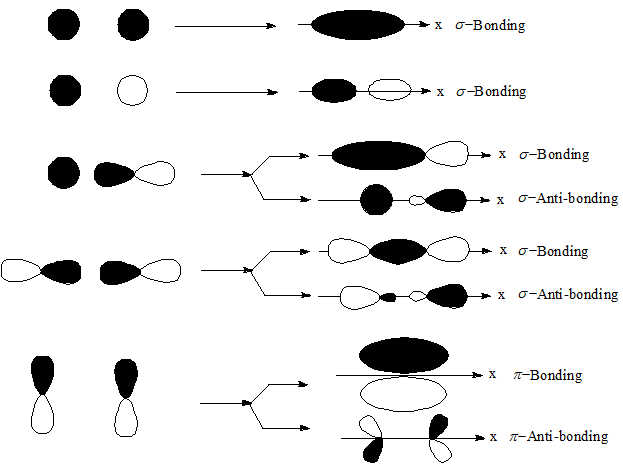 
Example: In hydrogen (H 2 ), the 1s orbital of each hydrogen atom overlap. Examples of sigma bond Sigma bond in Hydrogen This molecule has the two hydrogen atoms having 1 electron in an s orbitals. A sigma bond is always formed on the nuclear axis between two atoms. A sigma bond will always be a single bond. The s orbitals must be half-filled before they overlap. A sigma bond will be formed using electrons in s orbitals, p orbitals or hybrid orbitals. s-s Overlapping The s orbital from each atom participates in overlapping along the internuclear axis. They can be formed via the following combinations of atomic orbitals. Think of the first bond forming as a signal bond.\) molecule. Generally, all single bonds are sigma bonds. Both acquired their names from the Greek letters and the bond when viewed down the bond axis. Sigma bonds are formed by end-to-end overlapping and Pi bonds are when the lobe of one atomic orbital overlaps another. De a pie bond constricts rotation around the Sigma bond axis. Sigma and pi bonds are formed by the overlap of atomic orbitals. In order to form a pie bond, we need to have two regions of orbital overlap. The concept of sigma bonding is extended to describe bonding interactions involving overlap of a single lobe of one orbital with a single lobe of another. Yes, anything that forms a bond head on with one region of overlap is a sigma bond. Sigma bonds are obtained by head-on overlapping of atomic orbitals. True True/False: bonds formed from overlap of atomic s orbitals are always sigma bonds. Select A pi bond consists of This problem has been solved You'll get a detailed solution from a subject matter expert that helps you learn core concepts. True/False: A pi bond restricts rotation about the bond axis. Select v A pie bond restricts rotation about the sigma- bond axis. Bonds formed with atomic s Orbital's air. Select) Bonds formed from atomic s orbitals are always sigma bonds. Anything on top of that rpai bonds so sigma and a pie is a double a sigma and two pies is a triple next statement. The first bond a single bond is always Sigma. The next statement, a triple bond consists of one pie bond and two Sigma bonds. Explanation: Sigma Bond: This type of covalent bond is formed by the axial overlapping of half-filled atomic orbitals. POSSIBLE ORBITAL COMBINATIONS TO GENERATE SIGMA MOLECULAR ORBITALS For simplicity, if we examine only the s, p, and d orbitals, let's suppose that all orbitals we are examining are similar enough in energy to interact. The atomic orbitals overlap along the inter-nuclear axis and involve end-to-end or head-on overlap. A double bond is always a Sigma bond and a pie bond. bonds must be made by orbitals that overlap head-on. Explanation: Sigma Bond: This type of covalent bond is formed by the axial overlapping of half-filled atomic orbitals. the strongest bond, is formed by the head-on overlapping of atomic orbitals. Sigma bonds exhibit cylindrical symmetry to the internuclear axis. Exercise 13 oooo p orbital p orbital sigma bond s orbital p. The first statement is to Sigma bombs comprised a double bond thesis faults. The first covalent bond formed between two nuclei is always a sigma. Bonds formed from overlap of atomic s orbitals are always sigma bonds. This question is a Siris of true and false statements that you are asked to identify as true or faults associated with bond formation, pi bonds and the Sigma bonds. Question: A pi bond consists of two pairs of electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
